
Investigation of The Key Flavor Precursors in Chicken Meat
1. What is Flavor?
1.1 Key Flavor Compounds Identified in Chicken Meat
1.1.1 Differences between studies on key odor compound
1.1.2 Similarities between studies on key odor compound
1.1.3 Main compounds important for chicken flavor
1.2 Overview of Flavor Forming Reactions
1.2.1 Maillard Reaction & Strecker Degradation
1.2.2 Degradation of Thiamin
1.2.3 Oxidation of Lipids
1.3 Precursors of Meat Flavor
1.3.1 Ribonucleotides
1.3.1.1 Concentrations of ribonucleotides in chicken meat
1.3.2 Reducing and phosphorylated sugars
1.3.2.1 Concentrations of sugars in chicken meat and other meats
1.3.3 Amino acids
1.3.3.1 Sulfur-containing amino acids
- Role of cysteine
- Role of methionine
1.3.3.2 Concentrations of amino acids in meat
1.3.4 Thiamin
1.3.4.1 Concentration of thaiamin in chicken meat
1.3.5 Fatty acids and Lipids
1.4 Formation of Flavor Precursors in Meat Post-Slaughter
1.4.1 Post-mortem changes during conversion of muscle to meat
1.4.2 Glycogen pathway
1.4.3 ATP degradation pathway
![]()
1. What is Flavor?
In 1969, the U.S. Society of Flavor Chemists proposed the following definition: ‘Flavor is the sensation caused by those properties of any substance taken into the mouth which stimulates one or both senses of taste and smell, and also the general pain, tactile and temperature receptors in the mouth’ (Heath 1978). Flavor is a complex mixture of sensory input composed of taste (gustation), smell (olfaction) and tactile sensation of food as it is being chewed, a characteristic that the food scientists often term “mouthfeel’’. Scientists generally describe human taste perception in terms of four qualities: saltiness, sourness, sweetness and bitterness. Some have suggested, however, that other categories exist as well, most notably ‘umami’, the sensation elicited by glutamate, one of the 20 amino acids that make up the proteins in meat, fish and legumes (Smith and Margolskee 2001).
Odor, in contrast, has many different descriptors. The odor of a product is detected when its volatiles enter the nasal passage and are perceived by the olfactory system (Meilgaard et al. 1987). The odorous substances can be detected in the air above the food and in some case may dictate whether we decide to consume the food or not (Farmer 1999). The amount of volatiles which escape from a product is affected by the temperature and by the nature of the compounds (Lyman et al. 1982). Volatility is also influenced by the condition of a surface: at a given temperature, more volatiles escape from a soft, porous, and humid surface than from a hard, smooth, and dry one. The sorting of aroma sensations into identifiable terms continues to challenge sensory professionals (Meilgaard et al. 1987). The terminology in this field is very wide; according to Harper (1972) some 17000 odorous compounds are known and a good perfumer can differentiate 150 to 200 odorous qualities. Many terms may be ascribed to a single compound (thymol = herb-like, green, rubber-like), and a single term may be associated with many compounds (lemon = α-pinene, β-pinene, α-limonene, β-ocimene, citral, citronellal, linalool, α-terpenol, etc.).
Flavor is a very important component of the eating quality of meat and there has been much research aimed at understanding the chemistry of meat flavor which has resulted in the identification of over 1000 volatile compounds from cooked meats (Mottram 1991). A considerable amount of work, especially by the flavor companies, has been conducted and reviewed on the production of artificial flavorings to imitate cooked meat flavor (MacLeod and Seyyedain-Ardebili 1981). In recent thirty years, reviews have been written to explain how desirable flavor is generated (Thomas et al. 1971; Gordon 1972; Patterson 1974; Farmer 1992; Shi and Ho 1994; Mottram 1998; Farmer 1999).
A review of the flavor of poultry could cover a variety of aspects. In this introduction, the volatile compounds and precursors important for chicken flavor formation will be reviewed briefly. The quantities of the precursors present in chicken and their role for flavor formation are the major focus of this thesis and will be reviewed in more detail.
1.1 Key Flavor Compounds Identified in Chicken Meat
Aroma compounds in chicken are largely formed during the cooking process. Indeed, raw meat has none of the aroma of cooked meat, and the characteristic aroma is largely generated during cooking (Crocker 1948). In fact, the flavor compounds are generated via chemical reactions occurring between natural precursors present in raw meat during heating.
Since the desirable meaty flavor develops only after heating, the identification of these volatile compounds could lead to the identification of the possible chemical formation route and to achieving a better understanding of the components involved in these chemical reactions. It would then be possible to try to identify the limiting precursor or component leading to the formation of the characteristic volatile aromas of cooked chicken meat. Although, the cause of differences in the intensity of desirable flavor between chickens from different sources is not clear (Farmer 1999), different concentrations of important precursors in raw meat might be leading to variation in sensory quality of chicken meat.
Most research on the chemistry of flavor formation in poultry has been conducted on chicken (Gallus domesticus). Nevertheless, the principles behind the generation of flavor will be common to all species of poultry (Farmer 1999). Raw meat contains a number of natural components which participate in a series of chemical reactions during heating to generate a large number of volatile compounds responsible for the cooked meat aroma and flavor The primary reactions occurring upon heating that can lead to meat flavor include pyrolysis of amino acids and peptides, carbohydrate degradation, interaction of sugars with amino acids and peptides, degradation of ribonucleotides, and thermal degradation of lipids (McLeod and Ames 1987) as illustrated in Figure 1a.
Figure 1a: Chicken Flavor Formation
 |
|---|
Identification of the volatile compounds which give cooked poultry its desirable aroma may help to determine which chemical reactions are responsible for forming these compounds during cooking (Farmer 1999). Once these chemical pathways have been elucidated, it may be possible to investigate potential precursors involved in these reactions and deduce which of them in raw meat, are needed to produce the desirable poultry aroma (Farmer 1999).
About 500 volatile aroma compounds have been reported in chicken (Schroll et al. 1988; Schliemann et al. 1988; Maarse and Visscher 1989; Mottram 1991; Ramarathnam et al. 1991; Ramarathnam et al. 1993; Werkhoff et al. 1993) Many of these have relatively high odor thresholds and make little contribution to the overall flavor Others may be present at very low concentration but, due to their very low odor thresholds, have a very large effect on overall flavor (Farmer 1999).
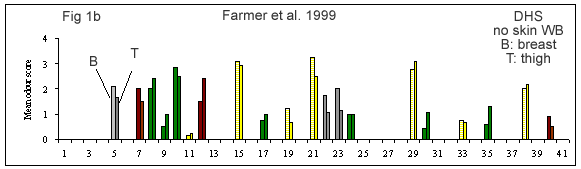
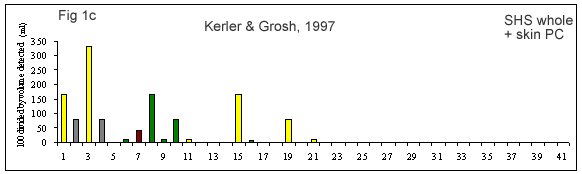
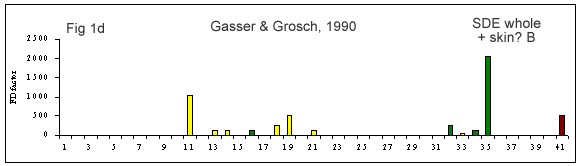
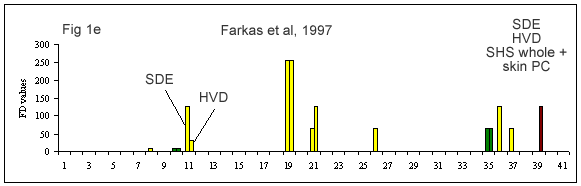
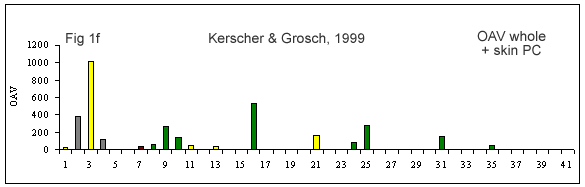
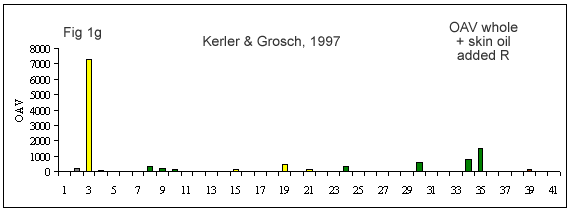
A few reviews on the volatile compounds contributing to poultry flavor have been published e.g. (Ramaswamy and Richards 1982; Wilson and Katz 1972; Steverink 1981; Farmer 1999). A number of research studies have attempted to determine which compounds are most important for the characteristic flavor or aroma of chicken (Gasser and Grosch 1990; Farkas et al. 1997; Kerler and Grosch 1997; Kerscher and Grosch 1999; Farmer et al. 1999).
Table 1A shows a summary of some important volatile compounds and their odor descriptors, derived from the studies cited above. A comparison of the aromagrams
(Figures 1b-g) derived from each of these studies shows that their results differ considerably. It is interesting to look at what has caused this diversity.
1.1.1 Differences between studies on key odor compound
First of all, the source and background of the chicken used for the studies cited in Table 1A and Figures 1 b-g is unknown. The type of sample used by these authors also varies considerably. Some included whole chicken (Figures 1c, d, e, f, g), breast or thigh (Figure 1b), with skin (Figures 1c, d, e, f, g) or without skin (Figures 1b, d). Comparison of breast versus thigh (Farmer et al. 1999) does not explain the differences between these data and those for whole chicken (Kerler and Grosch 1997). Presence of skin in the samples analysed in (Figures 1c-g) may explain the greater importance of lipid oxidation products (e.g., compound 35).
Table 1A: Odor descriptors and identities of some important volatile compounds
| No. |
LRI |
odor |
Probable identity |
Reference* |
|
|---|---|---|---|---|---|
| Non-polar |
Polar |
||||
|
|
|
|
|
|
|
| 1 |
<500 |
<500 |
S, putrid |
hydrogen sulphide |
b, d |
| 2 |
<500 |
<500 |
sweet |
acetaldehyde |
b, d |
| 3 |
<500 |
<500 |
S, putrid |
methanethiol |
b, d |
| 4 |
560 |
823 |
malty |
methylpropanal |
b, d |
| 5 |
- |
890 |
washing |
unknown |
e, |
| 6 |
700 |
925 |
green |
pentanal |
b, |
| 7 |
662 |
957 |
malty |
3-methylbutanal |
b, d |
| 8 |
802 |
1122 |
green, plastic |
hexanal |
b, c, d, e |
| 9 |
1005 |
1287 |
fatty |
octanal |
b, d, e |
| 10 |
978 |
1328 |
mushroom |
1-octen-3-one |
a, b, c, d, e |
| 11 |
875 |
1320 |
meaty |
2-methyl-3-furanthiol |
a, b, c, d, e |
| 12 |
923 |
1012 |
popcorn |
2-acetyl-1-pyrroline |
a, e |
| 13 |
898 |
998 |
S, catty |
3-mercapto-2-pentanone |
a, d |
| 14 |
995 |
1072 |
earthy |
trimethylthiazole |
a |
| 15 |
975 |
1432 |
gassy, metallic |
dimethyltrisulphide |
b, e |
| 16 |
1103 |
1394 |
fatty |
nonanal |
a, b, d |
| 17 |
- |
1398 |
mushroom |
1-nonen-3-one |
e |
| 18 |
968 |
1022 |
meaty |
2,5-dimethyl-3-furanthiol |
a |
| 19 |
- |
1429 |
roasted, coffee, meaty |
2-furanmethanethiol |
e |
| 20 |
- |
1447 |
sour |
acetic acid |
b |
| 21 |
903 |
1039 |
potatoes |
methional |
a,c, b, d, e |
| 22 |
- |
1542 |
potatoes |
unknown |
e |
| 23 |
- |
1523 |
stale, damp, potatoes |
unknown |
e |
| 24 |
1160 |
1260 |
cardboard-like |
(E)-2-nonenal |
a, b, d |
| 25 |
- |
|
fatty, green |
(Z)-2-nonenal |
c |
| 26 |
1097 |
1617 |
putrid |
3,5-dimethyl-1,2,4-trithiolane |
c |
| 27 |
780 |
1645 |
sweaty |
butyric acid |
b, d, c |
| 28 |
1020 |
1633 |
roasted, meaty |
2-acetylthiazole |
a, e |
| 29 |
- |
1680 |
chicken |
2-methyl-3-(methyldithio) furan |
e |
| 30 |
1215 |
1343 |
fatty, soapy |
(E,E)-2,4-nonadienal |
a, b |
| 31 |
- |
- |
cucumber |
(E,Z)-2,6-nonadienal |
d |
| 32 |
1350 |
1455 |
tallowy, sweet |
2-undecenal |
a |
| 33 |
1107 |
1241 |
roasty |
2-acetylthiazoline |
a, b |
| 34 |
|
ca 1800 |
fatty |
other 2,4-decadienal |
a, b, d |
| 35 |
1807 |
|
fatty, deep fried |
a, b, c, d |
|
| 36 |
1169 |
1776 |
putrid |
5,6-diH-2,4,6-triMe-4H-1,3,5-dithiazine |
c |
| 37 |
1200 |
1812 |
putrid |
5,6-diH-2,4,6-triMe-4H-1,3,5-dithiazine |
c |
| 38 |
|
1977 |
meaty, popcorn, S, chicken |
2-methyl-3-(methyltrithio) furan |
e |
| 39 |
1071 |
2037 |
caramel, sweet |
4-hydroxy-2,5-dimethyl-3(2H) furanone |
b, c, d |
| 40 |
1115 |
2209 |
hydrolysed protein |
3-hydroxy-4,5-dimethyl-2(5H)-furanone |
b |
| 41 |
1685 |
1898 |
tallowy, fruity |
γ-dodecalactone |
a |
The cooking method used by these authors also varied. Some chicken samples were cooked in a flask using a boiling water bath (Farmer et al. 1999), whereas, the use of pressure cooker at 116°C or 119°C, and roasting the whole chicken with coconut oil at 180°C, were preferred by Kerler and Grosch (1997) and Kerscher and Grosch (1999), respectively. The aroma of chicken will differ depending on the cooking method, but the four samples cooked by pressure cooker (Figures 1c, e, f and g) are not markedly similar.
The other divergence between studies carried out by these authors is their choice of extraction method. Static headspace (Kerler and Grosch 1997; Farmer et al. 1999) seems to be more sensitive to highly volatile compounds (compounds 1-10) than simultaneous distillation extraction (Gasser and Grosch 1990; Farkas et al. 1997) or high vacuum distillation (Farkas et al. 1997). High vacuum distillation and simultaneous distillation extraction give many similarities (compounds 11, 19, 21, 35, Fig 1e), but also differences for volatile compounds such as, 3-mercapto-2-pentanone, trimethylthiazole, nonanal, 2-undecenal (compounds 13, 14, 16 and 32, respectively) being detected only with simultaneous distillation extraction method (Fig 1d). In two studies (Kerler and Grosch 1997; Kerscher and Grosch 1999), quantities and odor thresholds were determined and odor activity values were calculated. Many methods of analysis failed to detect some volatile S-containing compounds (e.g. dimethylsulfide, 2-methyl-3-(methyldithio)furan, 2-acetylthiazoline).
In addition, since birds of different origin might have different concentrations of precursors and if different amounts of precursors can lead to different levels of each volatile compound, then, the lack of information concerning the origin of the birds and the concentration of natural precursors for each bird might explain a part of the diversity of their results (Figures 1b-g).
Compound type: |
|
|---|---|
1.1.2 Similarities between studies on key odor compound
General agreements between authors (Gasser and Grosch 1990; Kerler and Grosch 1997; Farkas et al. 1997; Kerscher and Grosch 1999; Farmer et al. 1999) were observed regarding the importance of compounds such as 1-octen-3-one, 2-methyl-3-furanthiol, 2-furanmethanethiol, methional (compounds 10, 11, 19 and 21, respectively) which were detected in all studies. Other compounds such as hexanal, (E,E)-2,4-decadienal (compounds 8 and 35, respectively) were present in four of five studies. Thus, it might be assumed that these compounds are important for chicken flavor, by whatever method these were determined.
1.1.3 Main compounds important for chicken flavor
The most important study on chicken flavor in recent years is probably the one published by Gasser and Grosch (1990). Using aroma extract dilution analysis, they identified 16 primary odor compounds in chicken broth. Fourteen of these compounds were structurally identified as 2-methyl-3-furanthiol, 2-furanmethanethiol, methional, 2,4,5-trimethylthiazole, 2-trans-nonenal, nonanal, 2-formyl-5-methylthiophene, p-cresol, (E,E)-2,4-nonadienal, (E,E)-2,4-decadienal, 2-undecenal, ß-ionone, γ-decalactone and γ-dodecalactone. 2-Methyl-3-furanthiol, identified in Gasser and Grosch (1990) as the most important flavor compound contributing to the meaty flavor of chicken broth, has also been recognized as a character impact compound in the aroma of cooked beef (Gasser and Grosch 1988) and canned tuna fish (Withycombe and Mussinan 1988). 2-Methyl-3-furanthiol and its oxidative dimer, bis-(2-methyl-3-furyl)disulphide, possessing characteristic meat flavor notes, have been found by Evers et al. (1976) among the volatile products from heating thiamine hydrochloride with cysteine hydrochloride and hydrolysed vegetable protein. These two compounds were also found in volatile products of thiamine degradation (van der Linde et al. 1979; Hartman et al. 1984). Thiamine has, therefore, been recognized as one possible precursor of the formation of the meaty aroma compounds, 2-methyl-3-furanthiol and bis-(2-methyl-3-furyl)disulphide. However, thiamine is not the sole source of 2-methyl-3-furanthiol. When ribose or 5’-inosine monophosphate (IMP) reacted with cysteine, a significant amount of 2-methyl-3-furanthiol and the bis-(2-methyl-3-furyl)disulphide was formed (Farmer and Mottram 1990; Grosch et al. 1990; Zhang and Ho 1991). However, the formation of 2-methyl-3-furanthiol from either ribose or IMP requires the participation of sulphur-containing amino acids, cysteine or cystine, or pepetide, glutathione (Shi and Ho 1994), whereas, thiamine does not require these components.
The importance of compounds, 1-nonen-3-one, 2-methyl-3-furanthiol, methional, 2-methyl-3-(methydithio) furan and 2-methyl-3-(methyltrithio)furan in increasing the sensory score for chicken aroma has been reported by Farmer et al. (1999). They also reported that the ‘roasted, coffee’ and ‘popcorn’ odors were due to 2-furanmethanethiol. They emphasised the importance of S-containing compounds which arise from the Maillard reaction between cysteine and/ or methionine and a 5-carbon reducing sugar or ribonucleotide, or from thermal degradation of thiamine.
The loss of meaty, chicken-like and sweet odor notes and the formation of green, cardboard-like, metallic off-odors in refrigerated storage and reheating of boiled chicken were attributed to increases in hexanal (sevenfold) and decreases in 2-furanmethanethiol and (E,E)-2,4-decadienal (both sixfold) by Kerler and Grosch (1997).
Farkas et al. (1997) reported that 2-furanmethanethiol, 3-(methylthio)propanal, 4-hydroxy-2,5-dimethyl-3(2H)-furanone, (E,E)-2,4-decadienal, 2-methyl-3-furanthiol and 2-ethyl-3,5-dimethyl pyrazine were the most potent odorants of the pressure-cooked hen meat.
1.2 Overview of Flavor Forming Reactions
The chemical reactions by which the volatile compounds responsible for chicken aroma and flavor are formed include the Maillard reaction and Strecker degradation, lipid oxidation and the degradation of thiamine. In this section a brief overview of each chemical reaction will be presented.
1.2.1 Maillard Reaction & Strecker Degradation
The Maillard reaction between reducing sugars and amino acids is known to generate flavors similar to those of cooked foods. The Maillard reaction is the reaction between an amino compound (amine, amino acid, peptide, or a protein) and a carbonyl group in a sugar. The Maillard reaction contributes to the formation of flavor in most cooked foods. Even in its simplest form, between one amino acid and one reducing sugar, the Maillard reaction yields more than one hundred volatile products (Salter et al. 1988). Although the reaction has been extremely studied and is the subject of many reviews (Hurrell 1982; Nursten 1986; Tressl et al. 1993; Mottram 1994), it is interesting to note that the mechanism proposed by Hodge (1953) still provides the basis for our understanding of the early stages of this reaction (Mottram 1998). The first step of the reaction is the formation of a N-aldosylamine, which involves an addition reaction between the carbonyl group of the open chain form of an aldosugar and the amino group of an amino acid, peptide or other compound with the primary amino group (Mottram 1994). The subsequent elimination of water and molecular rearrangement gives a 1-amino-1-deoxy-2-ketose (Amadori product). Figure 1h shows a simplified version of the formation of rearrangement products from Amadori compounds and their possible degradation by dehydration and retro-aldolization (fission).
Products of the Maillard reaction include aliphatic aldehydes, furfural, furfural derivatives, ketone (including α-dicarbonyls), alcohols, cyclic ethers, pyrroles, pyridines, pyrazines, oxazoles and oxazolines (Bailey 1988; MacLeod and Seyyedain-Ardebili 1981). Among these classes of compounds, pyrazines are well characterized as the compounds which directly contribute to the roast or smoky aroma. Pyrazines such as trimethylpyrazine and 2-ethyl-3,5-dimethylpyrazine were reported to have odor threshold values as low as 50 ng/1 air and 0.01 ng/1 air, respectively (Wagner et al. 1999).
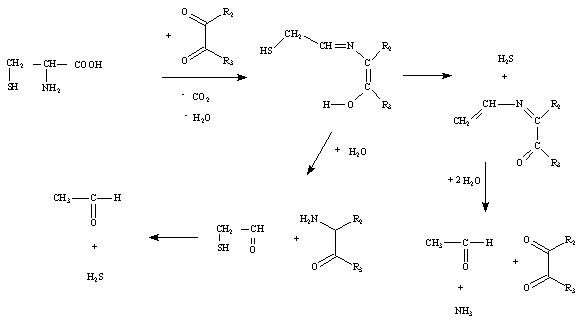
If sulphurous amino acids are involved, products may contain thiols, sulphides, thiophenes, thiazoles and thiazolines (MacLeod and Seyyedain-Ardebili 1981). Strecker degradation involves the oxidative deamination and carboxylation of an a-amino acid in the presence of a dicarbonyl compound (Mottram 1991). This leads to the formation of an aldehyde containing one fewer carbon atoms than the original amino acid, and an α-aminoketone. These aminoketones are important intermediates in the formation of several classes of heterocyclic compounds including pyrazines and thiazoles. In the Strecker degradation of cysteine, hydrogen sulfide, ammonia and acetaldehyde are formed, as well as the expected Strecker aldehyde mercaptoacetaldehyde and an α-aminoketone (Mottram 1991).
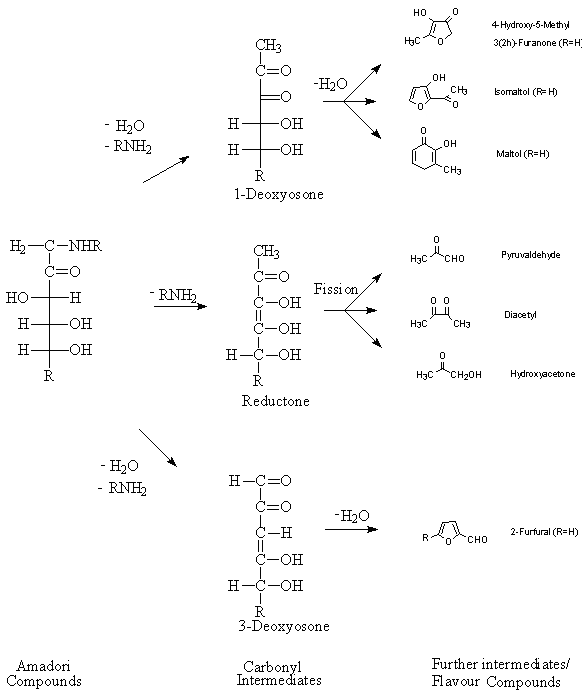
Figure 1i shows the primary products resulting from Strecker degradation of cysteine.
Figure 1i: Primary products resulting from Strecker degradation of cysteine (Kobayashi and Fujimaki 1965; MacLeod and Seyyedain-Ardebili 1981; Roos 1992)
Compound* |
Probable origin** |
3-mercapto-2-pentanonea |
Reaction of H2S with carbonyl compound from Maillard reaction1 |
Methionala, c, d |
Strecker degradation of methionine1 |
2-methyl-3-furanthiola, b, c |
Degradation of thiamine2,Maillard reaction between cysteine and ribose3 |
2,5-dimethyl-3-furanthiola |
Maillard reaction between cysteine and a hexose sugar |
2-furanmethanethiola |
Degradation of thiamine2, or Maillard reaction between cysteine and ribose4 |
2-formyl-5-methyl thiophenea |
Reaction between dicarbonyl compounds and H2S (both from Maillard reaction between amino acids and reducing sugars1 |
trimethylthiazole a |
From 2,3-butanedione, ethanal, NH3 and H2S (all from Maillard reaction)1 |
2,5(6)-dimethylpyrazined |
Strecker degradation of amino acids with dicarbonyl compounds (from Maillard reaction), and condensation of resulting amino-carbonyl compounds1 |
2-acetylpyrrolinea |
Maillard reaction between a reducing sugar and proline5 |
*References for the analysis of chicken aroma; a: (Gasser and Grosch 1990); b: (Werkhoff et al. 1990); c: (Farmer et al. 1999); d: (Siegl et al. 1995); **References for probable origin; 1: (Vernin and Parkanyi 1982); 2: (Güntert et al. 1992); 3: (van den Ouweland and Peer 1975); 4:(Farmer et al. 1989); 5:(Tressl et al. 1985)
Thiamine plays an essential role in many foods as a water-soluble vitamin. Additionally, its function as a flavor precursor in heated foods, e.g. meat, should not be neglected. However, certainly, this aspect depends very much on its amount and the specific conditions in the food system (Güntert et al. 1992). Another important field in which thiamine plays a remarkable role is the application of flavorings. Along with carbohydrates, amino acids, ribonucleotides, and other constituents, thiamine is widely used as a flavor precursor. This fact is clearly demonstrated by many patented reaction or processed flavors (Güntert et al. 1992). The thermal degradation of thiamine produces a number of compounds with particularly potent aromas, including furans, furanthiols, thiophenes, thiazoles, and aliphatic sulfur compounds, some of which have been reported in meat volatiles (Mottram 1991).
The role of thiamine as a potent flavor precursor is related to its chemical structure which consists of a thiazole as well as a pyrimidine moiety. The thermal degradation of this heterocyclic constituent leads to very reactive intermediates which are able to react directly to highly odoriferous flavor compounds or with degradation products of amino acids or carbohydrates (Güntert et al. 1992). Figure 1j shows some compounds formed by the thermal degradation of thiamine.
The thermal degradation of thiamine has long been a matter of analytical investigations. A comprehensive literature survey was published by Güntert et al. (1990). Studies on thiamine have resulted in the identification of 68 thermal degradation products (MacLeod and Ames 1986; Güntert et al. 1990). More than half contain sulphur and include aliphatic sulphides and thiols, sulphur-containing carbonyl compounds, sulphur-substituted furans, thiophenes, thiazoles, bicyclic compounds and alicyclic sulphur compounds. Many possess meaty aromas which are highly likely to contribute to the desirable flavor of cooked meat (Ames and Apriyantono 1992). However, a study of the possible routes of degradation of thiamine suggests that several compounds remain to be identified (Güntert et al. 1990).
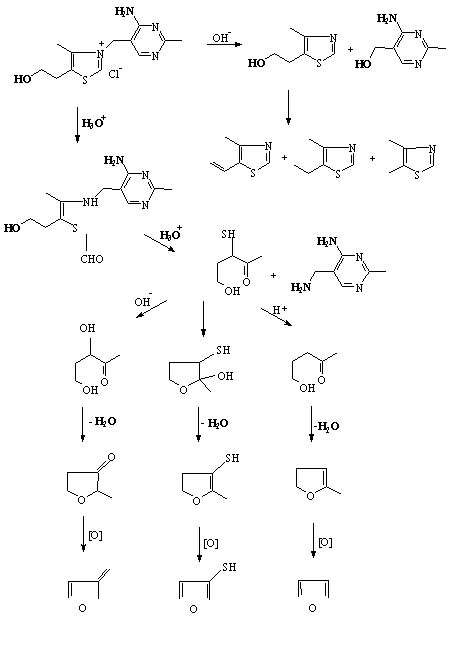
Grosch and co-workers (1992; 1993) studied the influence of thiamine as a possible precursor for the formation of the important meat flavor compound, 2-methyl-3-furanthiol, and its oxidised form bis-(2-methyl-3-furfuryl)disulfide. Van der Linde et al. (1979) found 5-(2-hydroxyethyl)-4-methylthiazole among the primary products of thiamine heated in buffer at 130°C, and suggested that it is formed as a result of the attack by a hydroxyl ion on the carbon atom connecting the thiazole and prymidine ring systems (Figure 1j).
Table 1C shows some important compounds from Table 1A, produced by thermal degradation of thiamine in chicken meat.
Table 1C: Important volatile compounds formed by thermal degradation of thiamine
Compound* |
Probable origin** |
2-methyl-3-furanthiola, b, c |
Degradation of thiamine1, Maillard reaction between cysteine and ribose2 |
2-furanmethanethiola |
Degradation of thiamine1, Maillard reaction between cysteine and ribose (or related compound)3 |
bis (2-methyl-3-furyl) disulphideb |
Degradation of thiamine1, Maillard reaction between cysteine and ribose followed by oxidationc |
*References for the analysis of chicken aroma; a: (Gasser and Grosch 1990); b: (Werkhoff et al. 1990); c: (Farmer et al. 1999); **References for probable origins; 1: (Güntert et al. 1992); 2: (van den Ouweland and Peer 1975); 3:(Farmer et al. 1989).
Lipid oxidation is well known as the cause of rancidity development but it can also contribute to desirable food flavors. This complex series of reactions is ubiquitious in the natural world and has been extensively studied and reviewed (Grosch 1982; Chan 1987). Table 1D indicates those which are derived from the thermal oxidation of lipids and which are believed to contribute to the flavor of chicken.
The volatile compounds derived from the thermal oxidation of lipids include aldehydes, ketones, and lactones with individual odors described as ‘green’, ‘fatty’, ‘mushrooms’ and ‘fruity’ (Farmer 1999).
Table 1D: Some important volatile compounds with lipid oxidation as origin
Compound* |
Probable origin** |
hexanala, d, e, f, g |
Thermal oxidation of linoleic acid |
nonanalb |
Thermal oxidation of n-9 fatty acids 1 |
trans-2-nonenalb |
Thermal oxidation of n-6 fatty acids 1 |
decanalb |
Thermal oxidation of n-9 fatty acids 1 |
2-undecenalb |
Thermal oxidation of n-9 fatty acids 1 |
γ-decalactoneb |
Oxidation of triglycerides 1 |
γ-dodecalactoneb |
Oxidation of triglycerides 1 |
(E,E)-2,4-nonadienalb |
Thermal oxidation of n-6 fatty acids 1 |
(E,E)-2,4-decadienal b, c, d |
Thermal oxidation of n-6 fatty acids 1 |
The major difference between the flavor of chicken broth and that of beef broth is the abundance of (E,E)-2,4-decadienal and γ-dodecalactone in the chicken broth. Both are well-known lipid oxidation products (Gasser and Grosch 1990). Carbonyl compounds formed by oxidative degradation of unsaturated lipids have been discussed by Minor et al. (1965) as a cause of the ‘chicken odor’ and an intensification of the ‘meaty odor’. In particular, the (E,E)-2,4-decadienal was found by Pippen and Nonaka (1957) to contribute to the aroma of chicken. Gasser and Grosch (1990) confirmed the importance of (E,E)-2,4-decadienal, with the highest FD factor of all the aroma compounds extracted from the chicken broth. They also reported that γ-dodecalactone and undecenal were potent odorants arising from a breakdown of lipids. The role of lipid-derived carbonyl compounds in poultry flavor has been extensively reviewed by Ramaswamy and Richards (1982).
A total of 193 compounds has been reported by Noleau and Toulemonde (1986) in the flavor of roasted chicken. Forty-one of them are lipid-derived aldehydes. The most abundant aldehydes identified in chicken flavor are hexanal and (E,E)-2,4-decadienal (Shi and Ho 1994). In view of the much lower odor threshold of (E,E)-2,4-decadienal (0.00007 mg kg-1) compared to hexanal (0.0045 mg kg-1) (van Gemert and Nettenbreijer 1977), the (E,E)-2, 4-decadienal should be the more important odorant for chicken flavor (Shi and Ho 1994). Hexanal and (E,E)-2,4-decadienal are primary oxidation products of linoleic acid. Cleavage of 13-hydroperoxide will lead to hexanal, and the breakdown of 9-hydroperoxide will lead to (E,E)-2,4-decadienal (Ho et al. 1989). Subsequent retro-aldolization of (E,E)-2,4-decadienal will produce 2-octenal and hexanal (Josephson and Lindsay 1987).
In beef and pork, the more saturated triacylglycerols played little part in the generation of volatile compounds or the characteristic odor of the cooked meat (Mottram and Edwards 1983). The more unsaturated phospholipids, present as components of the muscle cell walls, were of much greater importance for flavor development. Although triacylglycerols are therefore not needed for flavor development, they can affect juiciness (Keeton 1993) and the release of flavor (Chevance and Farmer 1997), and may influence how flavor is perceived.
1.3 Precursors of Meat Flavor
Meat tissue consists primarily of water, protein, fat, and carbohydrate, plus lesser amounts of nonprotein nitrogen-containing compounds and minerals and trace amounts of vitamins and other organic compounds (Mottram 1991). On heating, these components react to produce the complex volatile mixtures that are characteristic of meat aroma. A wide range of temperature conditions exist during normal cooking of meat; the centre of a rare steak may reach only 50°C, the centre of roast meat may attain 70-80°C, while the outside of grilled or roast meat will be subjected to much higher temperatures and localised dehydration of the surface will occur. It is not surprising, therefore, that a wide range of different flavor sensations are perceived in cooked meats. The nature and quantity of volatiles depend on the time and temperatures of heating. A number of these precursors may play a major role in formation of the volatile sulphur compounds important for roasted and meaty flavors (Mottram 1991). These include ribonucleotides, reducing and phosphorylated sugars, amino acids, thiamine and lipids which are briefly reviewed in this section.
1.3.1 Ribonucleotides
The naturally occurring ribonucleotides play an important role in the flavor of flesh foods (Kuninaka 1967; Jones 1969). Inosine-5’-monophosphate (IMP) and guanosine-5’-monophosphate (GMP) have been reported to enhance the meaty flavor and suppress sulphury, fatty, burnt, starchy, bitter and hydrolysed vegetable type flavors (Kuninaka 1967; Wagner et al. 1963). flavor enhancers, such as monosodium glutamate, inosine monophosphate and guanosine monophosphate are natural components of meat and are believed to make a major contribution to meat flavor (Farmer 1999). They have been shown to improve flavor and have been used by the Japanese for many years to give ‘umami’ (Reineccius 1994; Maga 1994). When using added IMP, Kurtzman and Sjostrom (1964) concluded that canned chicken-containing noodle soup was not flavor-enhanced. However, other products evaluated, including canned beef noodle soup, did show improvement with IMP addition.
At equimolar concentrations, IMP has most effect on aroma, but at concentrations related to those reported in the literature for red meat, ribose, IMP and glucose-6-phosphate were all found to increase ‘meaty’ and ‘roasted’ aromas (Farmer et al. 1996). Cambero et al. (2000) reported that flavor properties of beef broths were more influenced by the IMP concentration than guanosine monophosphate (GMP) and adenosine monophosphate (AMP) concentration and that small non-amino acid nitrogen compounds are more important than peptidic nitrogen with higher molecular weight than 600D. In addition to its flavor potentiating action IMP has also been reported in early studies to be essential for development of meaty aroma during cooking (Batzer et al. 1960; Koehler and Jacobson 1967). Farmer et al. (1996) reported that at four times (340 mg 100g-1) the reported concentration in beef and pork, IMP caused highly or very highly significant increases in both ‘meaty’ and ‘roasted’ aroma. They also reported that in most cases the addition of twice the reported concentration of IMP, gave no significant effect. Mottram and Madruga (1994) observed an increase in many sulphur-containing furans, including 2-methyl-3-(methyldithio)furan, on addition of IMP to beef at ten times its natural concentration. They affirmed that IMP was a precursor for 2-methyl-3-furanthiol and mercaptoketones, but 2-furanmethanethiol was not derived from IMP. IMP appears to act as a source of these thiols via the Maillard reaction and this reaction appears to involve the intermediate formation of 4-hydroxy-5-methyl-3(2H)-furanone and dicarbonyls, such as butanedione and pentanedione, and their subsequent reaction with hydrogen sulfide or cysteine (Mottram and Madruga 1994). IMP also appears to suppress the ‘green’ odor, probably caused by hexanal, and to reduce the formation of n-aldehydes and other lipid-oxidation products (Farmer et al. 1996).
Nucleotides and nucleosides are potential precursors of free ribose and ribose phosphate, which have been implicated in Maillard reactions during processing and storage of flesh (Tarr 1954; Macy et al. 1964). During thermal processing of flesh foods, IMP is degraded into inosine and hypoxanthine which have been reported to be either flavorless or bitter (Arya and Parhar 1979).
Macy et al. (1970b) reported that concentration of IMP decreases whereas that of free nucleosides and bases increases during roasting of beef, lamb and pork. Piskarev et al. (1972) reported that both AMP and IMP increase during thermal sterilisation of freshly slaughtered meat but decrease in aged meat. Suryanarayana-Rao et al. (1969) reported that IMP, inosine and hypoxanthine decrease during canning of shrimps but Hughes et al. (1966) and Mori et al. (1974) reported that hypoxanthine is stable at canning temperatures.
1.3.1.1 Concentrations of ribonucleotides in chicken meat
Literature provides a large number of methods of analysis for determination of ATP and its degradation products in muscle (Khan and Frey 1971; Shaw et al. 1979; Jeungling and Kammermeir 1980; Attrey et al. 1981; Kitada et al. 1983; Ryder 1985; Fujimura et al. 1995). A summary of different methods of analysis of ribonucleotides is discussed further in Section 2.3.1. Fujimura et al. (1995) has reported the concentrations of ATP breakdown products in chicken breast muscle (Table 1E).
Table 1E: ATP metabolites in chicken meat (Fujimura et al. 1995)
ATP metabolites |
Concentration (mg g-1 wet weight) |
IMP |
3.3 |
Inosine |
0.15 |
AMP |
0.10 |
ADP |
0.033 |
Hypoxanthine |
0.014 |
ATP |
0.012 |
1.3.2 Reducing and phosphorylated sugars
The important role played by ribose and cysteine in a model system leading to meat flavor formation, by heating a mixture of these compounds, was first investigated by Morton et al. (1960). The volatile compounds formed in the reaction between cysteine and ribose were dominated by sulphur-containing heterocyclic compound, particularly certain thiols and thiophenes (Farmer et al. 1989). They also reported 2-methyl-3-furanthiol and 2-furanmethanethiol as major volatile compounds from cysteine + ribose. Using a model system containing ribose and cysteine at pH 5.6, Mottram and Nobrega (1997) reported a large quantity of 2-furanmethanethiol with the dominant volatile in the ribose + cysteine system being 2-furfural. Reductions in the quantities of carbohydrates and amino acids in model systems, were observed during heating, the most significant losses occurring for cysteine and ribose (Mottram 1998). In meat, it has been proposed that ribose phosphate, from the ribonucleotides, is the principal precursor of furan and thiophenethiols (Mottram 1998). Dephosphorylation and dehydration of ribose phosphate form the important intermediate 4-hydroxy-5-methyl-3(2H)-furanone, which readily reacts with hydrogen sulfide (van den Ouweland and Peer 1975). Ribose-5-phosphate is formed from ribonucleotides such as inosine monophosphate, which is present in significant concentration in post mortem muscle (Patterson 1974; Macy et al. 1970a). Mottram and Nobrega (1997), reported a higher reactivity of ribose-5-phosphate compared to ribose when reacted with cysteine in a model system.
The dephosphorylation and dehydration of ribose-5-phosphate in aqueous solution, via 1-deoxypentosone, to yield 4-hydroxy-5-methyl-3(H)-furanone, has been previously reported (Peer and van den Ouweland 1968). The phosphorylation of ribose-5-phosphate may provide an easier route the furanone and diketone intermediates than the Maillard pathway, via Amadori intermediates,which is required for the free ribose system (Mottram and Nobrega 1997). As 2-furfural is formed via 3-deoxypentosone, which is produced from Amadori intermediates in the Maillard reaction, they also suggested that the formation of 2-furfural was not favoured in the ribose-5-phosphate system compared with ribose, as this compound was not produced by the dephosphorylation of ribose-5-phosphate.
Shibamoto and Russell (1976; 1977) heated glucose with NH3 then bubbled H2S through the solution. They reported that the volatiles resembled beef aroma. A methylene dichloride extract of the reaction mixture was separated by gas chromatography and 24 compounds reported in beef flavor studies were identified.
The addition of small quantities of ribose to raw beef has been shown to increase the quantities of key odor compounds, as well as meaty and roasted notes, in the cooked meat (Farmer et al. 1996; 1998). Ribose-5-phosphate also caused important changes while glucose and glucose-6-phosphate caused much smaller effects (Farmer et al. 1998). Hudson and Loxley (1983) reported that the addition of xylose to minced mutton increased the ‘mild’, ‘meaty’, ‘sweet’, aroma and flavor. In a model system, ribose-5-phosphate appears to be more reactive than ribose, producing much larger quantities of most volatile compounds responsible for meat flavor (Mottram 1998).
1.3.2.1 Concentrations of sugars in chicken meat and other meats
In order to determine the relative importance of these reducing sugars for flavor formation in meat, it is necessary, first, to know their natural concentrations. Very few studies on determination of sugars in chicken have been found in the literature. An investigation of sugars (but not their phosphates) was conducted by Lilyblade et al. (1962), using paper chromatography. The results of their studies are summarized in Table 1F.
Free sugars (glucose, fructose, ribose) and sugar phosphates (glucose-6-phosphate, fructose-6-phosphate, fructose-1,6-diphosphate) have previously been identified and quantified in aqueous beef extract using gas chromatography with complex derivatization procedures (Jarboe and Mabrouk 1974).
Compound |
Leg and thigh |
Breast |
||
|
Younga |
Oldb |
Young |
Old |
Inositol |
|
|
|
|
Freshc |
122 |
86 |
52 |
48 |
24 hour storage at 1°C |
148 |
120 |
72 |
72 |
Fresh |
135 |
117 |
54 |
57 |
6 day storage at 1°C |
188 |
147 |
80 |
82 |
|
|
|
|
|
Glucose |
|
|
|
|
Fresh |
188 |
155 |
141 |
105 |
24 hour storage at 1°C |
139 |
186 |
236 |
272 |
Fresh |
156 |
122 |
118 |
115 |
6 day storage at 1°C |
116 |
298 |
289 |
272 |
|
|
|
|
|
Keto sugar as fructose |
|
|
|
|
Fresh |
10 |
15 |
13 |
13 |
24 hour storage at 1°C |
22 |
24 |
44 |
42 |
Fresh |
15 |
8 |
13 |
9 |
6 day storage at 1°C |
22 |
39 |
50 |
40 |
|
|
|
|
|
Ribose |
|
|
|
|
Fresh |
1 |
1 |
3 |
<1 |
24 hour storage at 1°C |
4 |
3 |
5 |
1 |
Fresh |
1 |
1 |
1 |
1 |
6 day storage at 1°C |
9 |
6 |
14 |
7 |
Glucose and glucose-6-phosphate have also been quantified in beef by enzymic methods (Lawrie 1985). However, the reported concentrations can show considerable variation. For example, the concentration of ribose in beef is reported as 1 mg 100g-1 wet weight (Jarboe and Mabrouk 1974), 126 mg 100 g-1 wet weight (Cuzzoni and Gazzani 1984) and 524 mg 100 g-1 wet weight (Gazzani and Cuzzoni 1985). Table 1G summarises some results obtained for sugars in beef. It is unclear whether this variation is due to differences between meat samples or analytical methods. The natural quantity of ribose-5-phosphate in meat has not been reported.
Sugar |
Lyophilized |
Aqueous |
Adult |
Adult |
diffusatesa |
extractb |
animalc |
animald |
|
|
|
|
|
|
α-Glucose |
43.84 |
17.23 |
NR |
NR |
β-Glucose |
22.83 |
NR |
NR |
|
Fructose |
3.56 |
24.72 |
NR |
NR |
Ribose |
1.09 |
1 |
524 |
126 |
Glucose-6-phosphate |
NRe |
0.24 |
NR |
NR |
Fructose-6-phosphate |
NR |
0.27 |
NR |
NR |
Fructose 1,6-diphosphate |
NR |
0.76 |
NR |
NR |
1.3.3 Amino acids
Amino acids are important for the formation of meat flavor as they react with reducing carbohydrates via the Maillard reaction. The release of certain amino acids can also have an influence on meat quality attributes such as drip loss, water holding capacity (Lawrie 1992) and the development of flavor (Spanier and Miller 1993). Proteolytic degradation of myofibrillar proteins gives rise to increased substrate for enzymatic degradative systems with the concomitant release of free amino acids. It is possible therefore, that amino acid concentrations may also yield some information regarding the tenderness or flavor of meat (Lawrie 1992). On heating, protein and amino acids serve as a source of free ammonia. In addition, sulfur-containing amino acids, and proteins containing these amino acids, are precursors of hydrogen sulfide.
1.3.3.1 Sulfur-containing amino acids
Sulfur-containing volatile compounds are a major class of food aroma compound found in vegetables, cooked meat, and other processed foods (Gasser and Grosch 1988; Farmer and Mottram 1990; Block 1992).
- Role of cysteine
Cysteine is an important precursor for many sulfur-containing aroma compounds identified in meat and foods. Sulfur-containing flavors found in meat products are normally formed through thermal processing. It has been accepted that the sulfur-containing amino acids, cysteine and cystine, are indispensable components for generation of a meat-like aroma through thermal processing. (Shahidi et al. 1986; Tressl et al. 1989; Zhang and Ho 1991; Whitfield 1992). They participate in the Maillard reaction and Strecker degradation, it is also believed that upon heating, cysteine and cystine evolve hydrogen sulfide, one of the first compounds identified in early studies attempting to characterize the volatile compounds of cooked red meats (Crocker 1948) and poultry (Bouthilet 1951a; Pippen and Erying 1957). The small amount of hydrogen sulfide formed and volatilized when chicken is heated may be sufficient to influence aroma (Bouthilet 1951a; Pippen and Erying 1957). These sulfur-containing compounds are well recognized as major contributors to meat flavors (MacLeod 1986).
Mecchi et al. (1964) investigated the source of H2S in cooked chicken. They reported that the only sulfur compounds found in muscle nonprotein were methionine, taurine, and glutathione. Of these, only glutathione produced H2S. Therefore, the principal H2S precursor in chicken muscle nonprotein is sulfur occuring as cystine and/or cysteine in the glutathione. Muscle protein can also produce H2S and since methionine does not produce H2S, this sulfur must also come from cystine and/or cysteine (Mecchi et al. 1964). Glutathione gives H2S about 180 times as fast as chicken muscle protein, but, because there is approximately 1 to 2 thousand times as much protein as glutathione in muscle, protein is the principal H2S precursor (Mecchi et al. 1964). Glutathione can also take part in the Maillard reaction with reducing sugars (Mecchi et al. 1964). However, gluthatione reacts differently from cysteine. The cysteine residue is in the middle of the glutathione molecule. The amino group of the cysteine residue is bound in the peptide bond so that no Strecker degradation between the cysteine residue and dicarbonyl compounds can occur (Zheng et al. 1997).
Hydrogen sulfide release increases with the time and temperature of heating, presumably a result of protein denaturation and reduction of S-S bonds to –SH (Arnold et al. 1969).
Glutathione produces H2S at the beginning of the cooking, while the cysteine in muscle protein serves as the precursor of this compound in meat on prolonged heating (Ohloff et al. 1985). Since all proteinaceous foods probably emanate H2S upon heating, any effect on food flavor must be attributed to the concentration of H2S, the reaction of H2S with other compounds, or both (Zheng et al. 1997).
- Role of methionine
L-Methionine is one of the essential amino acids for man (Rose et al. 1955; Irwin and Hegested 1971). Methionine also has been widely used in the flavor and food industries to produce reaction flavors such as baked potato, fried potato, coffee, and meat flavors (Hertz and Shallenberger 1960; Ballance 1961; Hodge 1967; Chen 1968; Lee et al. 1973; Fan and Yeuh 1980; Silwar and Tressl 1989). The contribution of methionine to flavor formation was found to be mainly through thermal degradation or through thermal interactions with other food ingredients, especially reducing sugars. Herz and Shallenberger (1960) reported that heating methionine and glucose in an aqueous solution at 100°C or in mineral oil at 180°C would generate a potato aroma. El-Ode et al. (1966) indicated that heating methionine and sugars in water at 100°C yielded a cabbage-like aroma. Casey et al. (1965) reported that heating methionine was degraded by compounds such as glucose to produce methional, dimethyl sulfide, and dimethyl disulfide. Shigmatsu et al. (1977) reacted six sugars with methionine and cysteine. A mixture of equimolar amounts of sugar and amino acids was heated at 190°C for 15 min under reduced pressure. They reported that in most cases the main product was the Strecker aldehyde, methional, or products derived from this aldehyde, such as the corresponding alcohol. Besides dimethyl sulfides and methional, several volatile compounds were identified from the thermal degradation of methionine with or without glucose or dicarbonyl compounds (Fujimaki et al. 1969; Rijke et al. 1981; Ho et al. 1982; Hartman and Ho 1982; Tressl et al. 1989).
1.3.3.2 Concentrations of amino acids in meat
Free amino acids (nonprotein) and some peptides are very important substances, playing important roles eliciting characteristic tastes of foods (Nishimura and Kato 1988). Their direct participation in chemical reactions leading to the formation of volatile compounds responsible for the meat flavour has been well established. Better knowledge of the concentration of these amino acids is crucial to allow their relative importance for flavour formation to be determined.
Fujimura et al. (1995) and Perez et al. (1997) reported the results of free (nonprotein) and total amino acids in chicken breast, respectively (Table 1H).
Amino acid |
Free amino acids (mg 100g-1 wet weight)a |
Total amino acids (mg 100g-1 wet weight)b |
Lysine |
5.8 |
1958 |
Glutamic acid |
5.3 |
3362 |
Glycine |
4.2 |
1000 |
Threonine |
4.0 |
1030 |
Alanine |
3.6 |
1300 |
Proline |
3.4 |
896 |
Serine |
3.3 |
896 |
Cysteine/ cystine |
None reported |
181 |
Methionine |
2.9 |
481 |
Arginine |
2.4 |
1424 |
Tyrosine |
2.0 |
771 |
Aspartic acid |
2.4 |
2110 |
Leucine |
1.4 |
1740 |
Phenylalanine |
1.0 |
910 |
Valine |
0.7 |
1005 |
Histidine |
0.5 |
730 |
Isoleucine |
0.5 |
960 |
a:(Fujimura et al. 1995); b: (Perez-Llamas et al. 1997)
As expected, higher concentrations for all amino acids were reported by Perez et al. (1997) compared to those obtained by Fujmura et al. (1995). The presence of cysteine reported by Perez et al. (1997) and not by Fujmura et al. (1995) was also expected, as for the total amino acids all proteins were first hydrolysed. However, it is not clear whether the origin of cysteine detected by Perez et al. (1997) was proteins, glutathione or it was present as free (nonprotein) amino acid. The concentration of glutathione was not reported.
1.3.4 Thiamin
Thiamine (vitamin B1) is a bicyclic compound containing sulphur and nitrogen and its thermal degradation can yield a wide range of S- and N-containing volatile compounds many of which possess potent aromas (Buttery et al. 1984; Güntert et al. 1992). The role of thiamine in meat flavour formation was discussed previously in Section 1.3.2.
1.3.4.1 Concentration of thaiamin in chicken meat
The concentration of thiamine in chicken meat is reported in the literature. Ang and Moseley (1980) have reported the amount of thiamine in chicken leg meat as 0.85 ± 0.03 μg g-1, wet weight. Abdulrahman and Abdelbary (1993), have reported a similar results in light and dark muscles, 1.51 ± 0.09 and 1.92 ± 0.03 μg g-1 wet weight, respectively, from raw broiler chicken meat. Leonhardt and Wenk (1997) have published similar results in chicken breast and thigh: 1.4 μg g-1 wet weight for both, using the method described by Rettenmaier et al. (1979).
1.3.5 Fatty acids and Lipids
Meat lipids may be divided into two main groups; the storage lipids and the structural and metabolic lipids (Coxon 1987). The storage lipids are the triacylglycerols present in the fat cells of adipose tissue and intramuscular marbling fat. It is the major component of the fat cells, usually constituting in excess of 70% of the weight of adipose tissue. Structural lipids are present in all tissues, but in amounts generally less than 1% of the tissue, and metabolic lipids occur in equally low concentrations. The main structural lipids are the phospholipids, sphingolipids and cholesterol, whereas the commonest of the metabolic lipids are mono- and di-acylglycerols, non-esterified fatty acids and cholesterol esters (Coxon 1987).
Table 1I lists the fatty acid composition of the phospholipid and triglyceride fractions of chicken meat (leg and breast) (Pikul et al. 1984). It is clear that the higher the content of polyunsaturated fatty acids in the phospholipid fraction makes it highly susceptible to oxidation (Shi and Ho 1994). A higher amount of polyunsaturated fatty acids in the phospholipid fraction was observed in leg (43.5%) compared to the breast meat (41.4%).
Table 1I: Fatty acid composition of the phospholipid and triglyceride fractions of chicken meat (Pikul et al. 1984)
Chicken meat |
Lipid type |
Fatty acid composition (%) |
||
Saturated |
Monounsaturated |
Polyunsaturated |
||
Breast |
Triglyceride |
33.8 |
42.7 |
25.6 |
Phospholipid |
35.3 |
21.1 |
41.4 |
|
Leg |
Triglyceride |
33.0 |
42.3 |
24.8 |
Phospholipid |
39.1 |
16.4 |
43.5 |
|
Souza et al. (1999) reported the lipids and fatty acids composition and polyunsaturated fatty acids/ saturated fatty acids (PUFA/SFA) ratio of roasted chickens (breast, thigh and skin). Table 1J summarizes some of their results.
Pieces |
Lipids |
C14:0 |
C16:0 |
C16:1 ω-7 |
C18:0 |
C18:1 ω-9 |
C18:2 ω-6 |
C18:3 ω-6 |
C20:0 |
PUFA/SFA |
|
|
|
|
|
|
|
|
|
|
|
breast* |
1.65 |
0.53 |
20.03 |
4.33 |
7.62 |
35.69 |
26.16 |
1.62 |
2.22 |
0.91 |
breast** |
0.78 |
0.35 |
21.46 |
2.79 |
11.59 |
29.32 |
23.15 |
1.10 |
2.43 |
0.68 |
thigh* |
6.45 |
0.42 |
19.43 |
4.89 |
6.44 |
38.13 |
27.60 |
1.86 |
1.15 |
1.07 |
thigh** |
3.70 |
0.43 |
19.77 |
4.91 |
6.86 |
37.57 |
27.44 |
1.77 |
1.38 |
1.03 |
skin |
26.54 |
0.44 |
21.94 |
5.43 |
5.61 |
39.83 |
26.69 |
1.84 |
0.49 |
1.00 |
1.4 Formation of Flavor Precursors in Meat Post-Slaughter
1.4.1 Post-mortem changes during conversion of muscle to meat
Post-mortem changes that occur in the conversion of muscle to meat not only alter some of its biochemical and physical properties but also play an important role in improving its keeping quality and acceptability as food (Pearson and Young 1989). The nature of these changes and their consequences for meat have been reviewed (Greaser 1986; Pearson 1987). During the post-mortem aging period meat shows significant alteration in the level of numerous chemical components such as sugars (Lilyblade and Peterson 1962), organic acids (Bodwell et al. 1965), peptides and free amino acids (Parrish et al. 1969), and metabolites of adenine nucleotides such as adenosine triphosphate, ATP (Dannert and Pearson 1967; Davidek and Khan 1967). Many of the subsequent chemical changes are brought about by enhanced hydrolytic activity. These chemical modifications in the aging meat result in a pool of flavour compounds and flavour intermediates; the latter can react/interact to form additional flavour notes during cooking, e.g., sugars and amino acids react during heating to form Maillard products (Bailey 1988).
1.4.2 Glycogen pathway
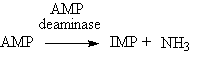
Glycogen is the main storage carbohydrate present in animal cells. It is a polysaccharide of D-glucose linked together by alpha-1,4 linkages, with each branch point, occurring about every 8-12 glucose residues, being in the alpha-1,6 configuration. Glycogen is hydrolyzed to glucose by the action of glycogen phosphorylase (Pearson and Young 1989). The major role of glycogen in post mortem muscle is release of glucose, which can be used to replenish the high-energy phosphate compounds. Thus, glycogen is largely degraded and is mainly responsible for the formation of lactic acid in muscle. Therefore, glycogen is ultimately responsible for the changes in the properties of muscle that accompany the drop in pH as glycolysis proceeds. After death, there are only a few sources of energy, such as the glycogen stores, residual ATP and ADP, and any unused creatine phosphate. As long as residual ADP sources remain high, the reaction below can also occur to provide additional ATP (Pearson and Young 1989). The AMP formed can then be deaminated to produce inosine monophosphate (IMP) and NH3.
![]()
1.4.3 ATP degradation pathway
The biophysical and biochemical changes that poultry muscle undergoes post mortem are, in general, the same as those reported for various mammalian species (De Fremery 1966). The most apparent change, of course, is the stiffening of muscle as it passes into rigor mortis. The chemical changes are (1) the disappearance of glycogen, adenosine triphosphate (ATP), and N-phosphorylcreatine, (2) the appearance of ammonia and IMP from the deamination of adenylic acid; and (3) the accumulation of lactic acid as a result of the anaerobic breakdown of glycogen. The accumulation of lactic acid lowers muscle pH from above 7.0 to ultimate values of 5.7 to 5.9.
At physiological death, cessation of blood circulation results in limiting the energy supply to the amount present in the muscles at that particular time as they are no longer capable of synthetising additional high-energy phosphate compounds or of removing the breakdown products from the system (Pearson and Young 1989). Thus, conversion of muscle to meat is delimited by the supply of glycogen, high-energy phosphates, and their metabolites present in the tissues at the moment of death.
Central to the process of conversion of muscle to meat under post mortem conditions is the role of the high-energy phosphate compounds and their metabolites ATP, ADP, creatine phosphate, and their metabolites are all involved in the energy transformations that occur in muscle (Pearson and Young 1989).
Figure 1k shows the breakdown of ATP in animals (Zubay 1983). In contrast to the catabolism of carbohydrates, lipids or amino acids, the catabolism of nucleotides results in no energy production in the form of ATP. In both GMP and AMP catabolism ribose-1-phosphate is released (Zubay 1983). In most meat, the sequence of nucleotide degradation follows a well-defined process: ATP; ADP; AMP; IMP; inosine; hypoxanthine; xanthine; uric acid (Figure 1k).
The concentrations of IMP in meat vary considerably between different animals and different muscles, and are affected by both pre- and post-slaughter production conditions (Mottram and Madruga 1994).
The predominant nucleotide in resting muscle is adenosine-5’-triphosphate (ATP). This undergoes enzymatic dephosphorylation to form adenosine-5’-diphosphate and then adenosine-5’-monophosphate (AMP). Deamination of AMP by the tissue enzyme AMP deaminase produces inosine-5’-monophosphate. These steps are fast and give rise to a rapid accumulation of IMP (Kennish and Krammer 1987). Satio and Arai (1959) found that these reactions took place rapidly during slow freezing of carp. Jones and Murray (1962) reported that trawled cod had little ATP and substantial amount of IMP at death compared with rested, well-fed fish.
The predominant nucleotide in resting muscle is adenosine-5’-triphosphate (ATP). This undergoes enzymatic dephosphorylation to form adenosine-5’-diphosphate and then adenosine-5’-monophosphate (AMP). Deamination of AMP by the tissue enzyme AMP deaminase produces inosine-5’-monophosphate.
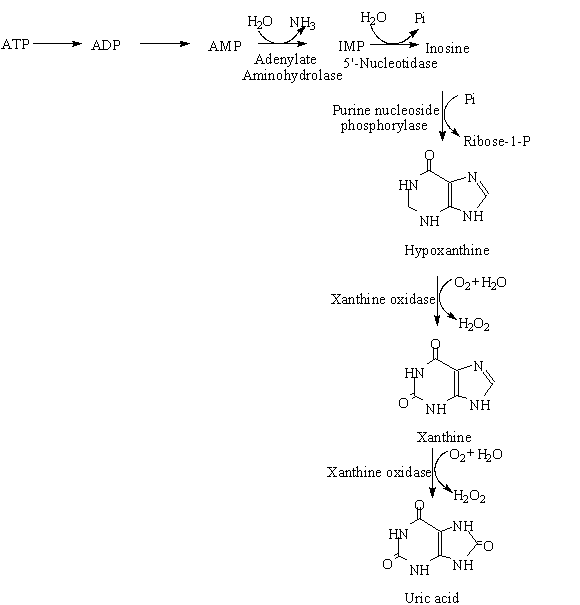
These steps are fast and give rise to a rapid accumulation of IMP (Kennish and Krammer 1987). Satio and Arai (1959) found that these reactions took place rapidly during slow freezing of carp. Jones and Murray (1962) reported that trawled cod had little ATP and substantial amount of IMP at death compared with rested, well-fed fish. The dephosphorylation of IMP to form inosine is carried out by the enzyme 5’-nucleotidase. The cleavage of inosine is performed by muscle ribosidase to form hypoxanthine and ribose (Kuninaka 1957).
Breakdown of IMP may also occur by the potential pathways proposed by Lee and Newbold (1963) which is shown in Figure 1l.
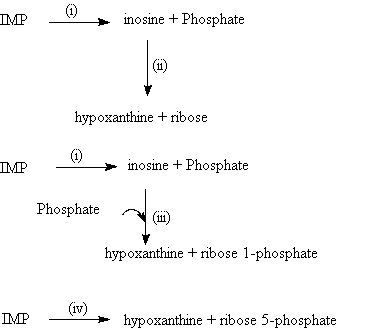
A comparison between Figures 1l and 1k shows that these authors (Newbold 1965; Zubay 1983) are proposing similar pathways for degradation of IMP into inosine, and also for the formation of ribose-1-phosphate from inosine. In contrast, the possible formation of ribose from inosine proposed by Lee and Newbold (1963) (Fig 1l) was not reported by Zubay
(Fig 1k).
In addition, the formation of ribose-5-phosphate (Lee and Newbold 1963) was reported to take place in liver and pancreas (Schmidt 1932; Ishikawa and Komita 1936). The real pathways for breakdown of IMP in chicken meat are not known. In addition, it is not clear from literature whether ribose-5-phosphate is formed post-slaughter or not. The formation of both ribose phosphates; ribose-1-phosphate and ribose-5-phosphate in chicken meat needs to be confirmed.
The optimum conditions for the enzymatic reactions occurring are not entirely elucidated. Lee and Newbold (1963) provided evidence using some enzyme inhibitors that, in ox muscle (longissimus dorsi), IMP was degraded to hypoxanthine by a pathway involving two steps. They reported that the presence of Cu2+ or Hg2+ inhibited the second step (ii) of the first reaction and inosine accumulated, while fluoride inhibited the conversion of IMP to inosine but not of inosine to hypoxanthine. They reported that in the presence of fluoride, there is no change in IMP concentration which indicated either IMP was not degraded through pathway (iii) or the splitting of IMP at the N-ribosidic link is completely inhibited by fluoride. However the second alternative seems to be unlikely since fluoride has been earlier shown by Lampen and Wang (1952) to have no inhibitory action on the hydrolytic cleavage of the N-ribosidic link of adenosine.
These authors also reported a marked effect of added phosphate on the formation of hypoxanthine from inosine and suggested that this step was phosphorylitic. On this basis, they concluded that the production of hypoxanthine from added IMP depends on phosphate for the phosphorolysis of inosine being provided by the dephosphorylation of IMP. That phosphate is required as a substrate for nucleoside phosphorylation and not for the activation or stabilisation of a hydrolytic nucleosidase is indicated by the finding that the hydrolases from yeast (Heppel and Hilmore 1952) and from Lactobacillus petosus (Lampen and Wang 1952) are not stimulated by phosphate.